 
When this reaction occurs, carbonic acid will develop, which has a formula of H2CO3. 
The water molecules that the carbon dioxide reacts with are the H2O in the equation. Carbon dioxide is the CO2 in the equation. Ocean acidification is a process that results from too much carbon dioxide being absorbed into the water. The equation itself is CO2 + H2O -> (H+) + (HCO3-). To best understand what ocean acidification is, it’s important that you know about the equation behind this process. Breaking Down the Ocean Acidification Equation This article talks about ocean acidification and how it directly impacts the environment as well as the life inside the oceans. The compound is then broken into hydrogen ions and bicarbonate, the former of which is what causes the oceans to become more acidic. When carbon dioxide reaches the ocean water, it forms what’s known as carbonic acid. While the process of ocean acidification can be difficult to understand, you can more readily know what it is by taking a look at the equation of CO2 + H2O -> (H+) + (HCO3-). This process is creating problems with the ocean ecosystems and the marine life that lives within the oceans around the world. While carbon dioxide is essential for natural processes like photosynthesis, ocean acidification is brought about when carbon dioxide is being created more rapidly than it can effectively be absorbed. Acidification occurs when excess amounts of carbon dioxide enter the atmosphere. Among the largest reasons for climate change is ocean acidification, which is causing oceans around the world to become more acidic than they’ve ever been.Įven if you’ve heard of ocean acidification, you may not know that the process doesn’t actually start in the ocean. These impacts extend from poor air quality to damaged habitats throughout the environment. All rights reserved.The impacts of climate change are already being felt all over the world. On behalf of the United States of America. Shall not be liable for any damage that may result fromįor NIST Standard Reference Data products. However, NIST makes no warranties to that effect, and NIST Uses its best efforts to deliver a high quality copy of theĭatabase and to verify that the data contained therein haveīeen selected on the basis of sound scientific judgment. The National Institute of Standards and Technology (NIST) Data from NIST Standard Reference Database 69:.Go To: Top, Reaction thermochemistry data, ReferencesĮnthalpy of reaction at standard conditions Kinetics and technological conditions for the synthesis of dimethyl carbonate,Ĭhem. Structural effects on rates and equilibria. Determination of the enthalpies of hydrolysis of some polyoxygenated hydrocarbons, Ī microprocessor-controlled system for precise measurement of temperature changes. Go To: Top, Reaction thermochemistry data, Notes 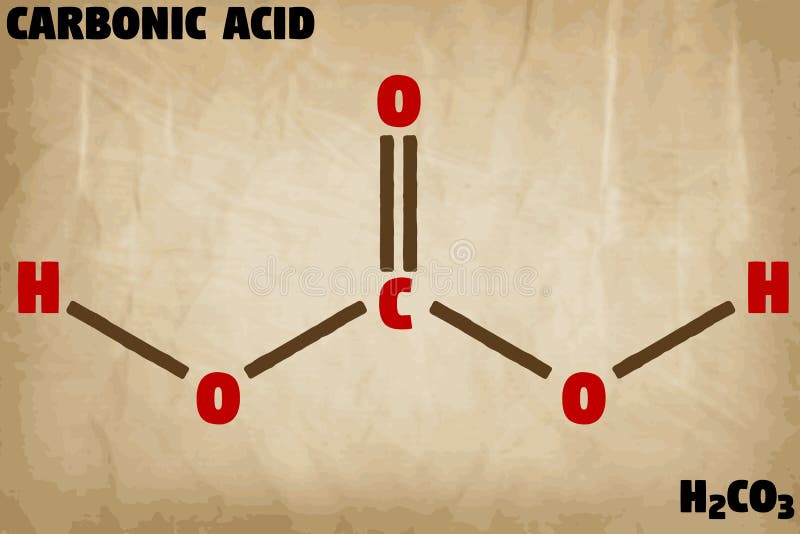
Reaction search pages in place of the enumerated reactionīy formula: C 5H 12O 4 + H 2O = C 3H 6O 3 + 2 CH 4O Quantityīy formula: C 4H 6O 3 + 2 CH 4O = C 3H 8O 2 + C 3H 6O 3 Quantity Secretary of Commerce on behalf of the U.S.A.ĭata compiled by: Hussein Y. Your institution may already be a subscriber.įollow the links above to find out more about the dataīy the U.S. With the development of data collections included in The purpose of the fee is to recover costs associated NIST subscription sites provide data under theĭata Program, but require an annual fee to access.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
